Research Papers using IBEX Heparinases
SARS-CoV-2 Infection Depends on Cellular Heparan Sulfate and ACE2
Thomas Mandel Clausen 1 et al
1 Department of Cellular and Molecular Medicine, University of California, San Diego, La Jolla, CA 92093 USA
in Brief:
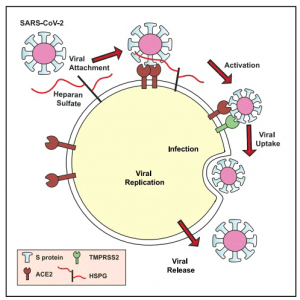 Clausen et al. provide evidence that heparan sulfate is a necessary co-factor for SARS-CoV-2 infection. They show that heparan sulfate interacts with the receptor-binding domain of the SARSCoV- 2 spike glycoprotein, adjacent to ACE2, shifting the spike structure to an open conformation to facilitate ACE2 binding.
Clausen et al. provide evidence that heparan sulfate is a necessary co-factor for SARS-CoV-2 infection. They show that heparan sulfate interacts with the receptor-binding domain of the SARSCoV- 2 spike glycoprotein, adjacent to ACE2, shifting the spike structure to an open conformation to facilitate ACE2 binding.
Highlights:
- SARS-CoV-2 spike protein interacts with heparan sulfate and ACE2 through the RBD
- Heparan sulfate promotes Spike-ACE2 interactio
- SARS-CoV-2 infection is co-dependent on heparan sulfate and ACE2
- Heparin and non-anticoagulant derivatives block SARSCoV-2 binding and infection
Heparinases from IBEX Pharmaceuticals were used in these experiments.
 Clausen et al. provide evidence that heparan sulfate is a necessary co-factor for SARS-CoV-2 infection. They show that heparan sulfate interacts with the receptor-binding domain of the SARSCoV- 2 spike glycoprotein, adjacent to ACE2, shifting the spike structure to an open conformation to facilitate ACE2 binding.
Clausen et al. provide evidence that heparan sulfate is a necessary co-factor for SARS-CoV-2 infection. They show that heparan sulfate interacts with the receptor-binding domain of the SARSCoV- 2 spike glycoprotein, adjacent to ACE2, shifting the spike structure to an open conformation to facilitate ACE2 binding.
IBEX heparinase III (50-012-001) used to elucidate the mechanotransduction pathways for the shear-induced nitric oxide production and to improve the understanding of the cardiovascular disease progression. This will enable the design of future therapeutics to rescue endothelial dysfunction.
Heparan sulfate proteoglycan glypican‑1 and PECAM‑1 cooperate in shear‑induced endothelial nitric oxide production.
Anne MarieW. Bartosch 1,2,3 Rick Mathews 1,4 Marwa M. Mahmoud 1 Limary M. Cancel 1 Zahin S. Haq 1 John M.Tarbell 1
1 Department of Biomedical Engineering, The City College of New York, 160 Convent Ave, New York, NY 10031, USA.
2 Department of Pathology and Cell Biology, Columbia University, New York, NY, USA.
3 Taub Institute for Research on Alzheimer’s Disease and the Aging Brain, Columbia University, New York, NY, USA.
4 The Knight Cardiovascular Institute, Oregon Health and Science University, Portland, OR, USA.
In this paper, researchers used IBEX Heparinase I, II and III to evaluate the immobilization of glycosaminoglycans (GAGs) using high salt. This study aimed at establishing a simple, efficient and low-cost in vitro method to study the biological roles of GAGs and their interactions with proteins.
A versatile salt-based method to immobilize glycosaminoglycans and create growth factor gradients.
Danique J. Hof 1 Elly M. M. Versteeg 1 Chris H. A. van de Lest 1,2 Willeke F. Daamen 1 Toin H. van Kuppevelt 1
1 Department of Biochemistry, Radboud Institute for Molecular Life Sciences, Radboud university medical center, PO Box 9101, 6500 HB Nijmegen, The Netherlands
2 Present address: Department of Equine Sciences and Department of Biochemistry and Cell Biology, Veterinary Medicine, Utrecht University, Utrecht, The Netherlands
IBEX Pharmaceuticals heparinase III used to demonstrate the regulatory role of Syndecan-3 in MSC adhesion and efficacy in inflammatory arthritis. The scholars used Hep III in digesting the Syndecan-3 in the cells and performing Western blot.
Syndecan‑3 regulates MSC adhesion, ERK and AKT signalling in vitro and its deletion enhances MSC efficacy in a model of inflammatory arthritis in vivo.
Fiona K. Jones 1,5 , Andrei Stefan 2,5 , AlasdairG. Kay 3 , Mairead Hyland 2 , Rebecca Morgan 2 ,
Nicholas R. Forsyth 4 , Addolorata Pisconti 1 & Oksana Kehoe 2
1 Department of Biochemistry and Cell Biology, Stony Brook University, Stony Brook, NY, USA.
2 School of Medicine, Keele University at the Robert Jones and Agnes Hunt (RJAH) Orthopaedic Hospital, Oswestry, UK.
3 Department of Biology, University of York, York, UK.
4 School of Pharmacy and Bioengineering, Keele University, The Guy Hilton Research Laboratories, Hartshill, Stoke on Trent, UK.
Nicholas R. Forsyth 4 , Addolorata Pisconti 1 & Oksana Kehoe 2
In this paper, the researchers tried to address one of the sourcing issues of Heparin from porcine origin by finding alternatives from other animal species, namely bovine and ovine sources. This paper was published in 2019. By using our Hep I enzyme, the scholars demonstrated that the ovine mucosal heparin has comparable anticoagulant and antiprotease activities to that of the porcine mucosal heparin and the bovine mucosal-derived heparin had somewhat lower potency than the above two heparins.
Comparative Pharmacological Profiles of Various Bovine, Ovine, and Porcine Heparins
Ahmed Kouta 1, Walter Jeske 1, Debra Hoppensteadt 1, Omer Iqbal 1, Yiming Yao 2, Jawed Fareed 1
1 Health Sciences Division, Cardiovascular Research Institute,
Loyola University of Chicago, Maywood, IL, USA
2 Ronnsi Pharma Co, Suzhou, China
Loyola University of Chicago, Maywood, IL, USA
In this paper researchers used IBEX Heparinase I and III enzymes to characterize the glycosaminoglycans (GAGs) in the velvet antlers of Sika deer. This study aimed at understanding the regulation of ossification mechanisms in deer antlers by GAG in the presence and absence of O- and N-sulfates. The ultimate goal is to determine the potential of GAGs as osteogenic agents for humans.
Compositional analysis of the glycosaminoglycan family in velvet antlers
of Sika deer (Cervus nippon) at different growing stages.
of Sika deer (Cervus nippon) at different growing stages.
Naoko Takeda-Okuda 1 , Shuji Mizumoto 2 , Zui Zhang 3 , Soo-Ki Kim 4 , Chi-Ho Lee 4 , Byong-Tae Jeon 5 , Yoshinao Z. Hosaka 6 , Kenji Kadomatsu 3 , Shuhei Yamada 2 , Jun-ichi Tamura 1
1 Department of Life and Environmental Agricultural Sciences, Faculty of Agriculture, Tottori University, Tottori 680-8551, Japan
2 Department of Pathobiochemistry, Faculty of Pharmacy, Meijo University, Tempaku-ku, Nagoya 468-8503, Japan
3 Department of Biochemistry, Nagoya University Graduate School of Medicine, Showa-ku, Nagoya 466-8550, Japan
4 Department of Animal Science and Technology, College of Animal Bioscience and Biotechnology, Konkuk University, Kwangjin-gu, Seoul 143-701, Korea
5 Korea Nokyong Research Center, Konkuk University, Chungju 380-701, Korea
6 Veterinary Anatomy, Joint Department of Veterinary Medicine, Faculty of Agriculture, Tottori University, Tottori 680-8553, Japan
IBEX Pharmaceuticals Heparinase III enzyme used in study of ultra-small gold nanorods (GNRs).
Cheng et al (2019) reported the fabrication of ultrasmall gold nanorods (GNRs), which are intended for use as the drug delivery vehicles to deliver the target drug into the endothelial cells. In this proof-of-concept study, the scholars have demonstrated that heparan sulfate is one component of the glycocalyx when chopped using IBEX Heparinase III enzyme, could increase the permeability of the nanorod into the endothelial cells. This research work is an essential first step towards uncovering the full potential of the ultra-small GNRs as drug delivery vehicles.
Ultrasmall gold nanorods: synthesis and glycocalyx-related permeability in human endothelial cells.
Ming J Cheng,1, Nandita N Bal,1, Priya Prabakaran,1 Rajiv Kumar,2,3 Thomas J Webster,1,4
Srinivas Sridhar,1,2 Eno E Ebong1,2,5
1Department of Chemical Engineering, Northeastern University, Boston, MA, USA
2Department of Physics, Northeastern University, Boston, MA, USA
3Millipore Sigma, Milwaukee, WI, USA
4Center of Excellence for Advanced Materials Research, King Abdulaziz University, Jeddah, Saudi Arabia
5Department of Neuroscience, Albert Einstein College of Medicine, New York, NY, USA
Srinivas Sridhar,1,2 Eno E Ebong1,2,5











